How Would You Determine Which Ph Indicator to Use
They are used to visually signal the acidity or alkalinity of an aqueous water-based solution. Before hand they will be introduced to two different types of indicators paper and liquid.

Make Your Own Ph Indicator Using Red Cabbage Science Experiments Science Experiments Kids Elementary Science Fair Experiments
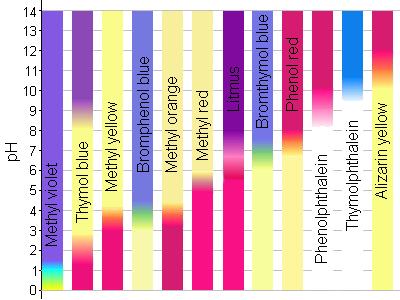
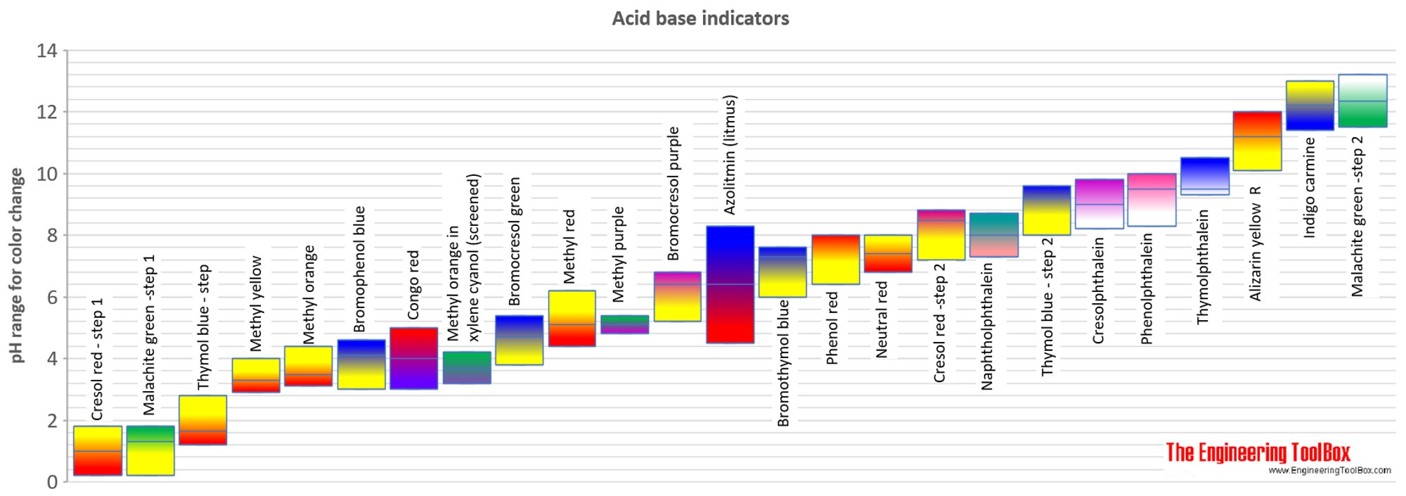
Bromocresol green is a pH indicator used to identify pH values between 38 and 54.

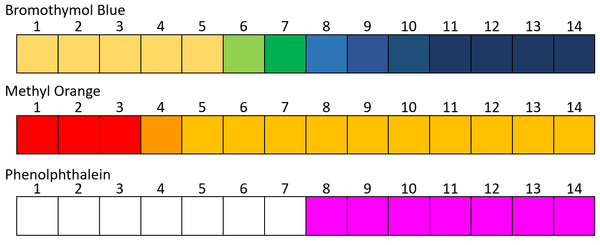
. Make sure you have taken at least 2 mL of juices in each test tube. Bromothymol blue It is a color indicator that turns yellow at acidic pH. Between pH 44 and 62 the indicator solution is orange.
Specifically they would use red and blue litmus paper phenolphthalein and bromothymol blue. We learned how to use the pH indicators and it was really fun to do that. The pH scale is used to determine the acidity or basicity of an acid or base.
Enter the pH ranges into the data table. The easiest part was checking the pH of the substances. Cassiemoreno18 cassiemoreno18 06032020 Chemistry College answered Use your Cabbage pH Indicator Key to determine the pH of each.
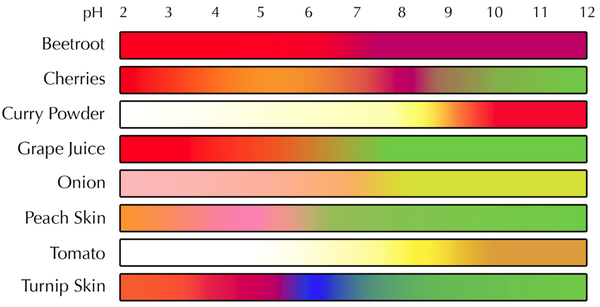
Classify the solutions as neutral acidic or alkaline. The photo on the right shows 12 solutions with cabbage indicator added. Then will go on to investigate the pH of 3 blood sample.
On the other hand pH at the equivalent point of acid-base react may not be 7. A pH indicator made up of several chemical compounds is known as a universal indicator. Each pH indicator has its own pH range for its color change.
The red cabbage can be made a textpH indicator by following these steps. Wash the pH probe with distilled water. PH Indicator Ranges Ascending pH Acid-base pH indicators are solutions that have a characteristic color at certain pH levels.
It means some indicators change their color in acidic rain pH 7 and some change in basic rain pH 7 Eg. Fill a blender halfway with hot water. Methyl red or bromcresol green will do for this one.
Alternatively you may dip the pH paper strips into each of the test solution. At high pH 62 and above the color is yellow. At low pH 44 and lower the indicator solution is red.
Take the given samples of fruit juices like lemon orange pineapple and apple in a separate test tube. I was thinking that you could set up a standard titration with ocean water as the unknown and drip the known HCl solution into it until the equivalence point but I dont know you would go about figuring out the equivalence point and consequently the pH of ocean water. At pH 75 or above bromthymol blue turns royal blue.
Use separate containers for each household solutionyou dont want to mix chemicals that dont go well together. The exact color you get depends on the pH of the water. Could you measure the pH of ocean water by using a strong acid-weak base titration.
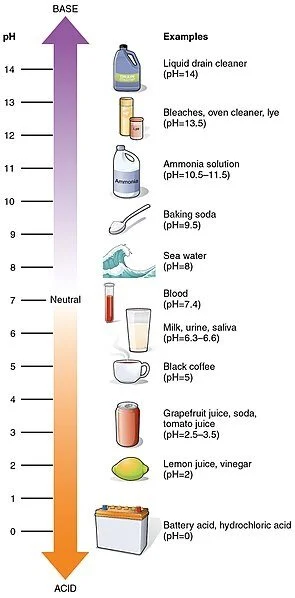
A solution with a pH of less than 7 is said to be acidic. Take a red cabbage and cut off about 2 3 cups. What we would probably change next time would be to organize better and write in a more organized way out.
After it is boiled turn the heat off and let it sit for ten minutes. Methylorange change its color at the pH range 3-45 and phenolphthalein change its color at the pH range 8-10. PH indicators generally change color over a range of two pH units.
To determine the pH of a solution using a pH indicator you need a. Use a separate dropper to. Knowing that the cabbage indicator always has the same color at a given pH value.
Enter the pH ranges into the data table. So the indicators you would choose would not be near any. This liquid is at about pH 7.
Begingroup ringo You want the indicator to change color after all the base has been converted distant from a ce pK_b not when it is being converted near a ce pK_b. How do you measure the pH of a solution. Compare the colour which appears on pH paper with the standard colour chart carefully.
Blend the leaves and water on high until the liquid turns purple and all the leaves are blended. Below pH 38 the. Directions for making red cabbage pH indicator.
They work only in colorless solutions in which a color change can be observed and can. At a neutral pH bromthymol blue is green. Lactose fermenting Yellow colonies and Lactose Non-fermenting colonies in CLED.
Methyl red is a pH indicator used to identify pH values between 44 and 62. You mix a drop of Universal indicator solution with approx 5 mL of the substance whose pH you want to knowYou can compare the color. Dont add too much.
Peel 3 or 4 big leaves off a head of red cabbage and chop the leaves into small pieces. Find an answer to your question Use your Cabbage pH Indicator Key to determine the pH of each. The pH of a solution can be estimated using universal indicator and a colour chart or measured using a pH meterThe pH of a solution can be measured using a pH meter.
2 See answers Advertisement. Compare the colour of the solution in the test tubes or on the pH papers with the standard colour chart to find the pH. To conclude this was a very interesting project.
How are acids and bases measured by a pH indicator. Using various concentrations of HCl and NaOH to measure a range of pH. Add the chopped cabbage leaves to the blender.
You will need an indicator that has a colour change very close to the desired pH. PH for Acids The pH of acids or acidic solutions is less than 7. As with precision test strips have less potential to be accurate compared with pH meters because of their relatively low resolution.
The indicators mentioned above show colour changes in the range of pH 50. What type of indicator can be used to determine the pH of a substance. Examples of pH Indicators.
The pH scale ranges from 0 to 14 on the scale. How to make the red cabbage a textPH indicator. The Procedure of Determination of pH Using Universal Indicator.
Add various household solutions to your indicator until it changes color. Pour about 50100 mL of your red cabbage indicator into each 250 mL beaker. First they would start by determining the pH of 6 known samples using all of the indicators.
PH indicators and their range pH indicators used in different culture media pH indicator. PH indicators are weak acids that exist as natural dyes and indicate the concentration of H H3O ions in a solution via color change. Record the reading on the pH meter.
When a test strip is used it changes color and is compared with a color chart. Range of acids and bases. In summary pH meters are usually much more precise and accurate than test strips.
There was nothing difficult in this experiment. Boil the pieces of red cabbage in a pot of water for a couple of minutes. Universal indicator can be used to determine the pH of a substance.
Using an existing scale to determine pH. A pH value is determined from the negative logarithm of this concentration and is used to indicate the acidic basic or neutral character of the substance you are testing. Put the end of the probe into the solution.

Making A Red Cabbage Ph Indicator Chemistry Lessons Teaching Chemistry Chemistry Experiments
At Home Stem Activities Make Your Own Ph Indicator Mcauliffe Shepard Discovery Center

Ph Indicators Biotechnology I Ppt Download

Diy Ph Indicator Eco Cleaning Products Switch Your Thinking

Acid Base Indicators Carolina Com

Free Download Of Student Worksheet For A Cabbage Ph Indicator Lab From Stemmom Org Teaching Chemistry Science Lessons Science Worksheets

Indicators Titration Mcat Content

Diy Ph Indicators Turn Your Kitchen Into A Chemistry Lab

Natural Ph Indicators Make A Ph Indicator Using Beetroot Or Cabbage

Preparation Of Indicator Solutions Pharmaceutical Guidelines Solutions Chemical Analysis Preparation
Informal Experiments How Do Ph Strips Work Precision Nutrition

Culture Media Ph Indicators Color Of Colonies Microbe Online



Comments
Post a Comment